Breakthrough Portable Diagnostic Device to Save Thousands of Lives in Low-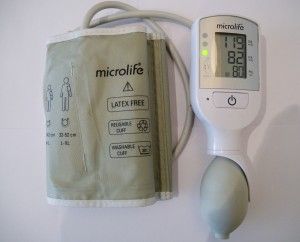 Resource Settings Resource Settings
With the introduction of the Sustainable Development Goals in 2015, the global community made a pact to dramatically reduce maternal mortality (MM) by the year 2030. Ongoing public health initiatives have led to an estimated 35% reduction in maternal deaths since the start of the millennium. But still there is much work to be done. In the year 2017, an average of over 800 women died each day (equaling 280,000 per annum) as the result of preventable causes related to pregnancy and childbirth. These deaths were highly concentrated in the low-resource settings of Sub-Saharan Africa and South Asia - in areas with limited access to vital health services and supplies.
Having witnessed the deadly implications of lacking diagnostic equipment during his years of work in Africa, Professor of Obstetrics Andrew Shennan, OBE, set out to address this problem. Through the support of the CradleTrial team at King’s College London and many generous sponsors – including the Bill and Melinda Gates Foundation with a contribution of $US1 million – Professor Shennan was able to begin development of a mobile diagnostic unit for the early detection of pregnancy and childbirth-related complications. Emphasizing accuracy, affordability and ease-of-use, the device would be targeted towards a reduction in the scandalously high rates of maternal mortality in many rural areas.
After 7 years of intense development at Kings College and St Thomas Hospital in London, this groundbreaking low-cost device – the Microlife Cradle VSA – is beginning to make an impact in reducing MM in low-resource areas around the world.
Over 800 Cradle VSA devices are in use in the Bidi Bidi and Nakivale refugee settlements in Uganda (250,000 inhabitants each), where they have been assisting in the rapid identification and prioritization of emergency cases for nearly two years. A successful pilot project in all hospitals in Freetown, Sierra Leone, also resulted in UK funding for a country-wide testing campaign there – the region with the worst MM record according to the WHO. And the Cradle VSA is used as an essential part of a VESPA (Vital signs alert Evaluating Shock index in Pregnancy Anemia) joint venture study in Karnataka State, India, examining the potential correlation between shock and anemia.
Key characteristics of the Cradle VSA include the highly accurate and sophisticated algorithm (six years in the making), the innovative and simple system for early warning sign detection and the low-power requirements. It can be charged with a standard USB phone charger or with a separately available solar charger for use in off-the-grid areas. Independent tests have also demonstrated an extremely robust built - the device maintains high accuracy after a fall from 20 meters, under conditions of extreme temperature and humidity, and even after over 20,000 uses. In recognition of the contribution made to solving urgent issues in the fields of health and development, the project team was awarded both the Newton Prize and PATH award.
This innovative $US 30 device offers an effective and affordable method for significantly and sustainably reducing preventable maternal deaths and is suitable for use in both small and large-scale Rotary projects in the field of Maternal and Child Health. The Cradle Trial VSA device is produced without profit and is available for non-profit users. Neither RFPD, nor any of the parties and organizations mentioned in this article have a financial stake in the production, promotion or sale of the Cradle Trial VSA.
Please visit the official CradlelTrial website at http://cradletrial.com/ to find out more. CradleTrial VSA orders can be directed to Dr. iur. Christoph von Luttitz, MBA (INSEAD), founder and President 2017/2018 of RC Mayfair (London) at CvL999@Gmail.com.
|
|